- My Forums
- Tiger Rant
- LSU Recruiting
- SEC Rant
- Saints Talk
- Pelicans Talk
- More Sports Board
- Fantasy Sports
- Golf Board
- Soccer Board
- O-T Lounge
- Tech Board
- Home/Garden Board
- Outdoor Board
- Health/Fitness Board
- Movie/TV Board
- Book Board
- Music Board
- Political Talk
- Money Talk
- Fark Board
- Gaming Board
- Travel Board
- Food/Drink Board
- Ticket Exchange
- TD Help Board
Customize My Forums- View All Forums
- Show Left Links
- Topic Sort Options
- Trending Topics
- Recent Topics
- Active Topics
Started By
Message
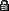
Sitting in an open thermos, which reaches room temperature (20°C) faster?
Posted on 6/1/21 at 5:35 pm
Posted on 6/1/21 at 5:35 pm
Posted on 6/1/21 at 5:39 pm to stendulkar
There have to be at least a couple of Keith Gonthier ME students here who paid attention in his Fluids / Heat Transfer class that should be able to answer this question. I personally have no idea whatsoever. 
Posted on 6/1/21 at 5:39 pm to stendulkar
39C water due to natural convection
Posted on 6/1/21 at 5:41 pm to stendulkar
quote:
Sitting in an open thermos, which reaches room temperature (20°C) faster?
Sir did you want that Baconator to be a combo or just the sandwich?
This post was edited on 6/1/21 at 5:58 pm
Posted on 6/1/21 at 5:42 pm to stendulkar
Regardless, my wife would simultaneously turn the downstairs AC on to cool the 39C water and turn the upstairs heat on to warm up the 1C water resulting in nothing but a massive power bill.
Posted on 6/1/21 at 5:50 pm to stendulkar
Same volume or same mass?
Even if you start with the same mass the 39 degree water will have a lower mass by the time it reaches 20 degrees due to increased evaporation even if it is minuscule.
Even if you start with the same mass the 39 degree water will have a lower mass by the time it reaches 20 degrees due to increased evaporation even if it is minuscule.
Posted on 6/1/21 at 5:51 pm to EngTiger
quote:
39C water due to natural convection
Now, qualitatively describe how the three heat transfer rates change over time as the two thermoses (thermi?) reach equilibrium with their surroundings. Assume the surroundings constitute an infinite heat sink or source and their temperature does not change.
Remember to phrase your response in the form of a question.
This post was edited on 6/1/21 at 5:54 pm
Posted on 6/1/21 at 6:17 pm to East Coast Band
Anything over 70 is now too hot over here.
Posted on 6/1/21 at 6:26 pm to stendulkar
the horse was named Friday
Posted on 6/1/21 at 6:26 pm to stendulkar
The thermos is the water's mother.
Posted on 6/1/21 at 6:27 pm to East Coast Band
quote:
Regardless, my wife would simultaneously turn the downstairs AC on to cool the 39C water and turn the upstairs heat on to warm up the 1C water resulting in nothing but a massive power bill.
Know the feeling.
Posted on 6/1/21 at 6:39 pm to stendulkar
is this while you are driving 100 mph?
Posted on 6/1/21 at 6:58 pm to stendulkar
I don't care for your punctuation. I immediately thought of sitting in a giant thermos.
Posted on 6/1/21 at 7:06 pm to HeadSlash
And the humidity
If humidity is 0% the 39C is faster due to evaporation
If it’s 100% humidity - the 1C will be faster due to condensation
And at a certain % humidity they would reach it at the same time
If humidity is 0% the 39C is faster due to evaporation
If it’s 100% humidity - the 1C will be faster due to condensation
And at a certain % humidity they would reach it at the same time
Posted on 6/1/21 at 7:12 pm to IT_Dawg
quote:
And the humidity
If humidity is 0% the 39C is faster due to evaporation
If it’s 100% humidity - the 1C will be faster due to condensation
And at a certain % humidity they would reach it at the same time
I like the depth you're bringing, but what kind of crappy thermos is allowing condensation? Should we assume it does the only thing it's supposed to do well? I would assume heat transfer only occurs on the surface if I were to make assumptions here.
Posted on 6/1/21 at 7:45 pm to calcotron
quote:
I like the depth you're bringing, but what kind of crappy thermos is allowing condensation? Should we assume it does the only thing it's supposed to do well? I would assume heat transfer only occurs on the surface if I were to make assumptions here.
You don’t think water can condense on the surface?
Posted on 6/1/21 at 8:16 pm to Christopher Columbo
quote:
The thermos is the water's mother.
Post-Vatican II, can a Catholic man marry his widow's sister?
Posted on 6/1/21 at 8:21 pm to IT_Dawg
quote:
And the humidity
I'm assuming no heat exchange with the thermos and the surroundings. So it's just out the top.
I would think the boundary layer would be thinner for the warm water because of natural convection mixing the air. Additionally while evaporation probably isn't the most important part, you'd get more evaporation off the warm water. So the mass of the warm one would be a little less over time.
So the hot one seems like the right answer, but it depends on the assumptions you make about the system.
Popular
Back to top

 11
11











